
There is a conversation happening in perimenopause clinics that focuses almost entirely on oestrogen — but leaves out the role of cortisol, stress, and inflammation in making menopause symptoms worse. This article addresses what gets left out.
This is not wrong. But it is significantly incomplete.
Because sitting alongside the hormonal story is a second story that most women in perimenopause are never told — and that, once understood, explains why some women experience the transition so much more intensely than others eating similar diets and living similar lives.
That story is about cortisol. About what happens when the stress hormone and the sex hormones occupy the same biological territory. About how chronic stress does not merely make you feel worse — it chemically amplifies every symptom you are already experiencing through a mechanism that is specific, well-documented, and directly responsive to the food and lifestyle choices you make every single day.
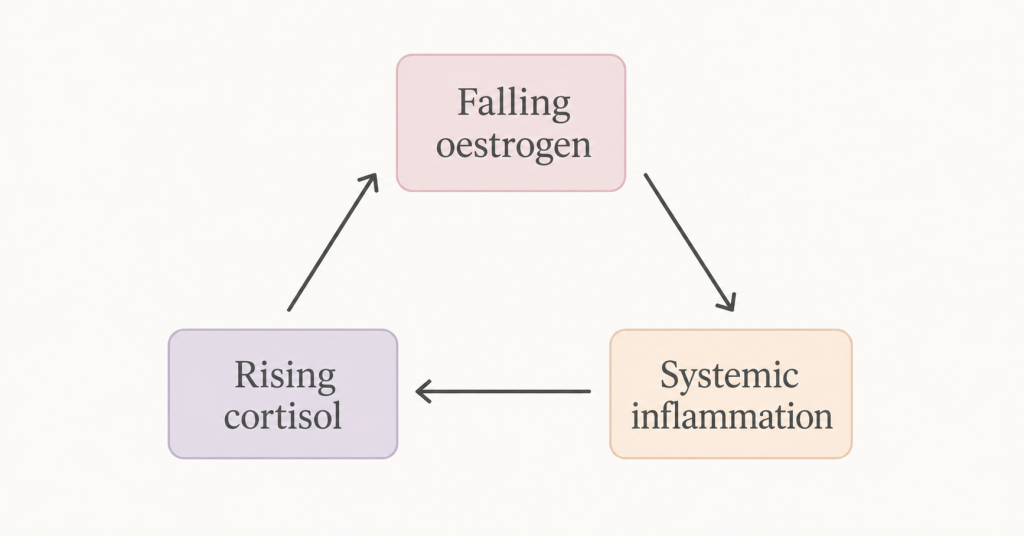
This is the menopause triangle: oestrogen decline, cortisol elevation, and systemic inflammation. Three forces operating simultaneously, each one amplifying the others. Understanding how they interact is one of the most useful things you can do for your experience of this transition.
Episode: “Cortisol and Menopause — The Triangle Nobody Explains” — Real Food Science Podcast
Key Takeaways
- Cortisol and oestrogen compete for the same biological raw materials — chronic stress accelerates the hormonal decline of perimenopause through a mechanism called pregnenolone steal
- Cortisol directly activates the same inflammatory pathways that falling oestrogen is no longer suppressing — creating a compounding inflammatory effect
- The HPA axis becomes hypersensitised during perimenopause — meaning stress responses are more intense, more prolonged, and harder to switch off than in earlier decades
- Hot flashes, joint pain, sleep disruption, weight gain, brain fog, and mood instability all have a cortisol component that sits alongside the oestrogen component
- Cortisol responds to dietary and lifestyle change faster than almost any other hormone — measurable changes within days, not months
- The food-first cortisol strategy targets blood glucose stability, omega-3 support, magnesium repletion, and the evening cortisol cutoff
The Biological Triangle — How Three Systems Collide
To understand why stress hits so differently in perimenopause, you need to understand how oestrogen, cortisol, and inflammation relate to each other — not as separate systems but as an interconnected triangle where each corner affects both others.
Corner one: Falling oestrogen
Oestrogen is the anti-inflammatory buffer that most women carry silently through their twenties and thirties — suppressing inflammatory cytokines, maintaining gut lining integrity, modulating immune activity, and protecting the brain, joints, and cardiovascular system from inflammatory damage. As it declines in perimenopause, this buffer withdraws. Inflammatory baseline rises. Every inflammatory stimulus — including cortisol — produces a more significant response than it did before.
Corner two: Rising cortisol baseline
Perimenopause does not directly cause chronic stress — but it reliably coincides with the life stage at which many women carry the heaviest stress burden. Career demands, parenting teenagers, caring for ageing parents, relationship pressures, financial concerns — the forties and early fifties are frequently described by women as the most demanding decade of their lives. Chronic stress keeps cortisol elevated continuously rather than in healthy, episodic spikes.
Beyond life circumstances, the HPA axis itself becomes more reactive during perimenopause. Oestrogen moderates the HPA stress response — it helps cortisol return to baseline after a stressor. As oestrogen falls, this dampening effect is lost. Stress responses become more intense, peak higher, and persist longer. The same stressor that produced a manageable cortisol response at 38 may produce a significantly larger and more prolonged response at 46.
Corner three: Systemic inflammation
Cortisol, paradoxically, is both anti-inflammatory and pro-inflammatory. In acute, short-term bursts it suppresses inflammation — this is why cortisol-derived steroids like prednisolone are used as anti-inflammatory medications. But in the chronically elevated state of ongoing stress, this relationship inverts. Cells become desensitised to cortisol’s anti-inflammatory signal — a process called glucocorticoid resistance — and the immune system loses its cortisol-mediated braking mechanism. Inflammatory cytokines rise. Gut permeability increases. The systemic inflammatory state that oestrogen was previously suppressing now has two drivers operating simultaneously.
The triangle is self-reinforcing: falling oestrogen increases HPA reactivity, which elevates cortisol, which drives inflammation, which further sensitises the HPA axis, which produces more intense stress responses, which produces more cortisol. Understanding this loop is the first step to interrupting it.
Pregnenolone Steal — How Stress Accelerates Hormonal Decline
This is the mechanism most women are never told about — and it directly connects the stress response to the rate of hormonal decline in perimenopause.
Pregnenolone is the precursor hormone from which your body manufactures cortisol, oestrogen, progesterone, testosterone, and DHEA. It is produced from cholesterol in the mitochondria of adrenal cells and is the raw material for the entire steroid hormone cascade.
Your body has a clear hierarchy of priorities in how it allocates pregnenolone. Cortisol sits at the top — because from your body’s evolutionary perspective, managing an acute threat is more urgent than maintaining sex hormone levels. When the stress signal is chronic and sustained, the enzyme systems that convert pregnenolone toward cortisol are preferentially upregulated. The pathways toward oestrogen and progesterone receive less substrate.
The result — called pregnenolone steal or cortisol steal — is that chronic stress depletes the precursor material available for sex hormone production. Oestrogen and progesterone levels drop faster and further than they would under the same hormonal trajectory without chronic stress. The perimenopause transition becomes more severe, more symptomatic, and more prolonged.
This mechanism has a specific implication that matters practically: reducing chronic stress in perimenopause is not just about feeling calmer. It is about slowing the rate of hormonal decline. Every sustainable cortisol-reduction strategy — dietary, behavioural, or both — is also a partial hormonal protection strategy.
Progesterone is particularly vulnerable to pregnenolone steal because it sits in a different branch of the steroid synthesis pathway from cortisol and is less prioritised when resources are scarce. This explains why the first hormonal symptoms of perimenopause — irregular periods, worsened PMS, sleep disruption, anxiety — are often progesterone-related symptoms, years before oestrogen begins its significant decline.
How Cortisol Specifically Amplifies Each Menopause Symptom
Understanding the cortisol-symptom connections in concrete terms makes the clinical picture much clearer than the generic “stress makes everything worse” framing.
Hot flashes
The hypothalamic thermostat that regulates the thermoneutral zone — the temperature window within which your body does not trigger a vasomotor response — is directly sensitised by both inflammatory cytokines and cortisol. Elevated cortisol activates the corticotropin-releasing hormone (CRH) pathway in the hypothalamus, which in turn modifies norepinephrine signalling in the temperature regulation circuits. Multiple studies have found that women with higher cortisol levels report significantly more frequent and more intense hot flashes, independent of their oestrogen status. Women who implement cortisol-reduction strategies consistently report meaningful reductions in vasomotor symptom severity within two to four weeks.
Sleep disruption
Cortisol follows a healthy diurnal rhythm — high in the morning to facilitate waking and energy mobilisation, declining steadily through the day, reaching its nadir around midnight. Chronic stress flattens and dysregulates this curve. Evening cortisol stays elevated, making it difficult to fall asleep. Blood glucose drops at 2-3am trigger emergency cortisol release to restore glucose — this is the mechanism behind the 2-3am waking that is almost universal in perimenopausal women with high stress loads. Poor sleep then raises next-day cortisol by 15-30% — completing the loop that keeps so many women in a state of exhausted but wired sleeplessness.
Abdominal weight gain
Cortisol drives fat storage in visceral adipose tissue — the metabolically active fat that accumulates around the abdomen. This is a direct hormonal effect rather than a caloric one: cortisol activates lipoprotein lipase in visceral fat cells and directs free fatty acids preferentially toward abdominal storage. It also promotes insulin resistance, which raises fasting insulin, which further promotes fat storage. The midlife abdominal weight gain that most women attribute entirely to oestrogen decline has a significant cortisol component — which is why dietary approaches that reduce inflammatory load and stabilise blood glucose produce better body composition results in perimenopausal women than simple calorie restriction.
Joint pain
Cortisol — paradoxically, given its anti-inflammatory pharmaceutical uses — promotes joint inflammation in the chronic state through glucocorticoid resistance and elevated IL-6 production. Joint cartilage is directly affected by sustained cortisol elevation, which suppresses the chondrocytes responsible for cartilage maintenance and repair. The morning stiffness that many perimenopausal women experience reflects overnight cortisol dysregulation as much as oestrogen-related joint changes.
Brain fog
Chronic cortisol elevation directly impairs hippocampal function — the brain region most involved in working memory and new learning. Extended glucocorticoid exposure reduces hippocampal volume in animal models and correlates with cognitive impairment in human studies. For perimenopausal women experiencing word retrieval difficulties, poor concentration, and slow processing speed, the cortisol contribution to their cognitive symptoms is often as significant as the oestrogen contribution — and considerably more directly addressable through lifestyle.
Mood instability
Cortisol drives tryptophan metabolism away from serotonin production and toward the kynurenine pathway — the inflammatory arm of tryptophan metabolism. Less serotonin means reduced mood stability, increased anxiety, and impaired emotional regulation. This is the neurochemical mechanism behind stress-related depression — the same pathway that connects gut permeability to mood through the LPS-cytokine cascade described in the leaky gut article.
The Food-First Cortisol Strategy
Cortisol is uniquely responsive to dietary and lifestyle intervention — faster than any other hormone in the menopause context. Here are the five dietary strategies with the strongest evidence for lowering cortisol specifically in perimenopausal women.
Strategy 1: Blood glucose stability is the master lever
The single most impactful dietary cortisol intervention is flattening the blood glucose curve. Every blood glucose spike followed by reactive hypoglycaemia triggers a cortisol response — your body treats glucose deficiency as a physiological threat and mobilises cortisol to restore circulating glucose. In perimenopausal women, where declining oestrogen has already reduced insulin sensitivity, these spikes are larger and the cortisol responses are more pronounced.
The practical implementation is covered in detail in the morning habits article, but the core principle is protein and fat at every meal, no meal gap longer than four hours, and elimination of the refined carbohydrates and sugars that produce the most dramatic glucose excursions. This single change consistently produces the fastest subjective improvements in stress resilience and cortisol-related symptoms.
Strategy 2: Omega-3 fatty acids directly reduce the cortisol response
EPA and DHA — the long-chain omega-3 fatty acids in oily fish — have been shown in multiple controlled trials to directly blunt the cortisol response to psychological stressors. A 2010 study in Psychosomatic Medicine found that omega-3 supplementation at 2.5g EPA/DHA daily produced a 19% reduction in cortisol response to a standardised stress test. The mechanism involves modulation of the HPA axis at the pituitary level and reduction of the neuroinflammation that sensitises the stress response.
For women whose primary fat sources are seed oils — which compete with omega-3s for incorporation into cell membranes — the cortisol-sensitising effect of chronic omega-6 overload is compounding their stress response throughout the day. The fat swap covered in the cooking fats article is therefore simultaneously an anti-inflammatory and a cortisol-reduction intervention.
Strategy 3: Magnesium is the cortisol brake
Magnesium has a specific inhibitory relationship with the HPA axis — it acts at multiple points in the cortisol production pathway to dampen the response to stress. Magnesium deficiency removes this inhibition and produces a hyperreactive stress axis. Up to 70% of perimenopausal women are deficient in magnesium, partly through dietary insufficiency and partly because cortisol itself depletes magnesium — creating a bidirectional relationship where stress depletes the mineral that would otherwise moderate the stress response.
The highest dietary magnesium sources are dark leafy greens, pumpkin seeds, dark chocolate, avocado, almonds, and legumes. Magnesium glycinate supplementation at 300-400mg before bed is the most well-absorbed form and consistently produces improvements in sleep quality and cortisol regulation within one to two weeks in deficient individuals.
Strategy 4: The evening cortisol cutoff
Cortisol cannot decline adequately in the evening while the nervous system is receiving threat signals. Every news notification, work email, difficult conversation, and stressful screen content after 8pm sends a low-level threat signal that blunts the natural evening cortisol decline. For perimenopausal women whose evening cortisol curve is already dysregulated, this input is particularly disruptive.
The food component of the evening protocol is complex carbohydrates at dinner — sweet potato, lentils, wholegrain rice, or oats — which raise brain serotonin over two hours as they digest. Serotonin is then converted to melatonin, supporting natural sleep onset. The behavioural component is the hard 8pm boundary for stress inputs, combined with chamomile or ashwagandha tea, which have specific GABA and cortisol-modulating effects respectively.
Strategy 5: Anti-inflammatory eating as HPA protection
The systemic inflammation driven by seed oils, emulsifiers, alcohol, and ultra-processed foods directly sensitises the HPA stress axis — making it more reactive to the same stressors. This is the dietary mechanism of stress amplification: a high-inflammatory dietary pattern does not merely worsen symptoms in isolation; it makes your stress response worse, which worsens your cortisol load, which worsens your inflammation, which worsens your symptoms.
The anti-inflammatory dietary approach — replacing seed oils, increasing diverse plant fibre, adding fermented foods, prioritising oily fish — is therefore simultaneously a cortisol management strategy. The food changes that reduce hot flashes and bloating are the same changes that produce a calmer, more manageable stress response. This is why the 30-Day Anti-Inflammatory Workbook structures each week around a specific aspect of this interconnected picture rather than treating inflammation as separate from stress and hormones.
The Lifestyle Layer — What Food Cannot Do Alone
Cortisol management cannot be achieved through food alone — and it is important to be honest about this rather than overstate the dietary case.
Sleep is non-negotiable. Poor sleep raises next-day cortisol by 15-30%. No dietary intervention compensates adequately for chronic sleep deficiency. The sleep strategies covered in the cortisol and sleep article — and the Cortisol & Sleep Reset Workbook — address this as the primary non-dietary priority.
Movement type and timing matter. Gentle to moderate exercise — particularly walking — lowers cortisol and is one of the most evidence-backed non-dietary cortisol interventions available. High-intensity exercise raises cortisol acutely and should be scheduled before 3pm to allow the cortisol spike to resolve before the evening window. The timing of exercise matters more than most women realise.
The parasympathetic nervous system needs deliberate activation. In a perimenopause body with a sensitised HPA axis, the nervous system defaults more readily to sympathetic dominance. Deliberate parasympathetic activation — through extended-exhale breathing, slow gentle movement, time in nature, or genuine relaxation (not passive screen consumption) — is a physiological necessity rather than an optional wellness practice. The three-breath technique before opening the phone, covered in the morning habits article, is one accessible entry point.
The stress load itself may need addressing. Food and lifestyle strategies optimise your physiological response to a given stress load. If the stress load itself is unsustainable — an impossible work situation, a relationship in crisis, a caregiving burden without support — optimising the physiological response has limits. This is not something any dietary approach can fix, and it deserves honest acknowledgement.
The 30-Day Structure — Why This Takes a Month
The cortisol triangle cannot be resolved in a week. The pregnenolone steal reverses gradually as the chronic stress signal diminishes. Glucocorticoid sensitivity takes two to three weeks to begin restoring. The gut permeability driven by elevated cortisol takes four to six weeks of combined dietary and stress management intervention to meaningfully improve.
This is why the 30-Day Anti-Inflammatory Workbook is structured as a monthly programme rather than a shorter intervention. Week one addresses blood glucose stability and the immediate dietary disruptors of cortisol regulation. Week two deepens the anti-inflammatory approach and introduces the gut healing layer. Week three addresses the sleep and evening cortisol protocol. Week four focuses on consolidation, stress resilience, and the sustainable habits that carry the improvements beyond the programme.
The workbook gives you the daily structure, the week-by-week science, and the symptom tracking system that makes your progress visible rather than invisible — because without tracking, the gradual improvements in cortisol regulation, sleep quality, and symptom burden accumulate below the level of conscious awareness until you compare day one to day thirty and see the full picture of what has changed.
Free Resource: Identify Your Personal Triggers
Understanding which dietary and lifestyle inputs are most directly driving your cortisol load helps you prioritise the changes with the greatest personal impact.
→ Download the free Menopause Bloat Trigger Checklist — a five-minute checklist that identifies your personal food and lifestyle inflammation triggers, including several that directly feed the cortisol-inflammation loop.
Go Deeper: Feel Like Yourself Again — The 30-Day Workbook
The 30-Day Anti-Inflammatory Workbook is the structured, guided approach to everything covered in this article — daily pages, weekly science deep-dives, symptom tracking across all four symptom categories, and the daily reflections that turn knowledge into lasting habit. Built specifically for women in perimenopause and menopause who want to understand what they are doing and why — not just follow a list of instructions.
FAQ
If I am on HRT does managing cortisol still matter? Yes — significantly. HRT addresses the oestrogen component of the menopause triangle but does not address the cortisol or inflammation components. Many women on HRT find that residual symptoms — persistent sleep disruption, ongoing brain fog, mood instability, abdominal weight gain — are primarily cortisol and inflammation driven rather than oestrogen driven. The cortisol management strategies in this article are complementary to HRT and specifically address the symptoms that HRT alone often does not fully resolve.
Can adaptogens like ashwagandha genuinely help? Ashwagandha — specifically the KSM-66 extract studied in clinical trials — has the most robust evidence of any herbal adaptogen for cortisol reduction. A 2019 study in Medicine found that KSM-66 at 300mg twice daily produced a 27.9% reduction in serum cortisol over eight weeks compared to placebo. This is a meaningful effect size. The evidence for other adaptogens — rhodiola, holy basil, eleuthero — is considerably thinner. Ashwagandha is the one worth considering if dietary and lifestyle strategies have been implemented and additional support is wanted. Discuss with a healthcare provider if you have a thyroid condition, as ashwagandha can interact with thyroid medication.
I am already eating well — why are my cortisol-related symptoms still severe? If diet is genuinely clean and cortisol symptoms persist, the remaining contributors are most likely: the stress load itself being unsustainable at a life circumstances level, sleep quality being insufficient to allow HPA recovery, subclinical thyroid dysfunction driving HPA hyperactivity, or the hormonal component requiring medical support. Dietary approaches optimise your physiological resilience — they cannot fully compensate for an unsustainable external environment or an underlying medical condition.
How quickly will I notice a difference if I implement these strategies? Blood glucose stabilisation produces noticeable changes in energy and stress reactivity within three to five days of consistent implementation. Magnesium repletion typically produces improvements in sleep quality and morning calmness within one to two weeks. The deeper cortisol curve normalisation — less 2-3am waking, reduced hot flash frequency, improved mood stability — typically emerges at three to four weeks of sustained approach. Tracking symptoms from day one makes this progression visible.
Is the 2-3am waking definitely cortisol and not menopause night sweats? Both can contribute and they frequently occur together, making them difficult to distinguish without tracking. The most reliable distinguishing feature is whether the waking is accompanied by sweating (more likely the vasomotor component) versus occurring with a clear, alert, anxious quality with no sweating (more likely the cortisol-glucose mechanism). Many women experience both patterns on different nights. The dietary strategies for both overlap significantly — blood glucose stability and the evening cortisol protocol address the cortisol component, while the anti-inflammatory approach addresses the inflammatory component of vasomotor symptoms.
Sources
- Tsigos, C. & Chrousos, G.P. (2002). Hypothalamic-pituitary-adrenal axis, neuroendocrine factors and stress. Journal of Psychosomatic Research, 53(4).
- Epel, E. et al. (2000). Stress and body shape: stress-induced cortisol secretion is consistently greater among women with central fat. Psychosomatic Medicine, 62(5).
- Kumari, M. et al. (2009). Association of diurnal patterns in salivary cortisol with all-cause and cardiovascular mortality. Journal of Clinical Endocrinology & Metabolism, 96(5).
- Kiecolt-Glaser, J.K. et al. (2010). Omega-3 supplementation lowers inflammation in healthy middle-aged and older adults. Brain, Behavior, and Immunity, 26(6).
- Pickering, G. et al. (2020). Magnesium status and stress: the vicious circle concept revisited. Nutrients, 12(12).
- Woods, N.F. et al. (2006). Cortisol levels during the menopausal transition and early postmenopause. Menopause, 13(5).
- Avis, N.E. et al. (2001). Is there an association between menopause status and sexual functioning? Menopause, 8(5).
